HeartMate 3
Respects the blood
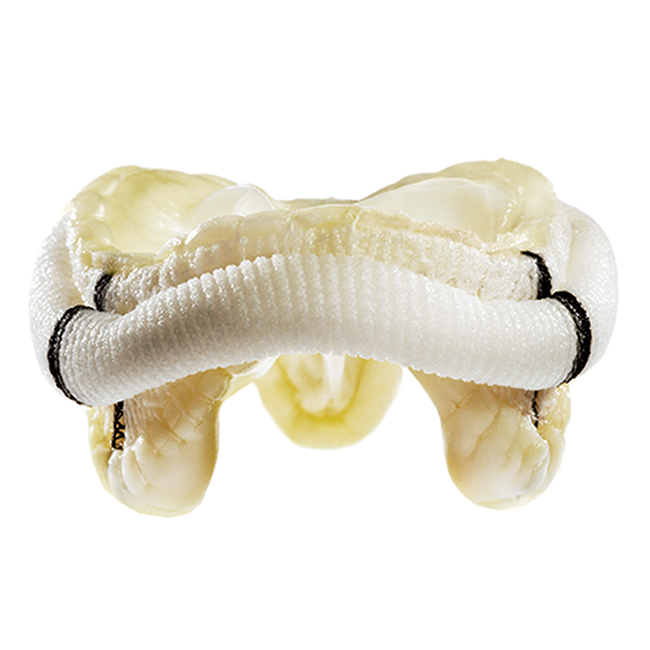
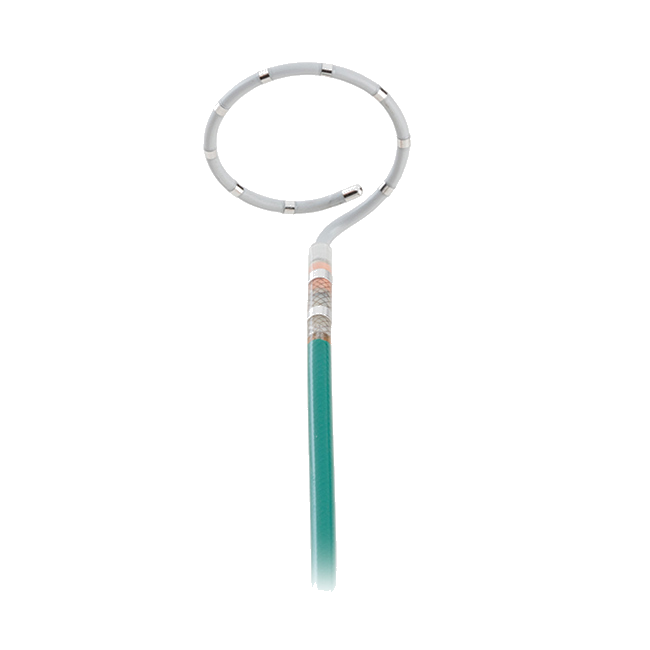
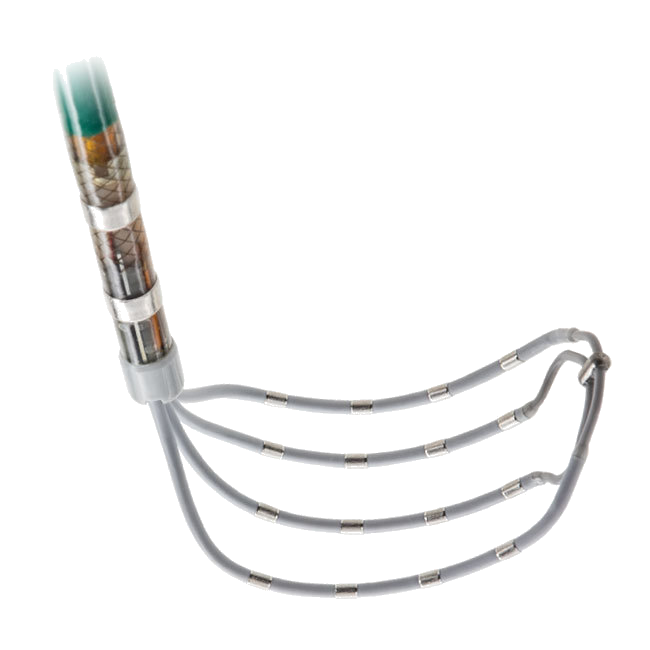
Building on the legacy of proven performance established by HeartMate II®, HeartMate 3 is designed to offer unprecedented blood-handling characteristics through the incorporation of Full MagLev™ flow technology.
Full MagLev flow technology allows the device’s rotor to be magnetically levitated, or suspended, by magnetic forces. The resulting contact-free environment is designed for hemocompatibility, with large, consistent blood-flow pathways designed to reduce blood shearing and hemolysis and to minimize complications.
Results from the HeartMate 3 CE Mark Clinical Trial may have significant implications for the care of patients with advanced heart failure:
- No pump thrombosis, hemolysis, or pump malfunctions or exchanges 36
- 98% 30-day survival and 92% 6-month survival (Excellent 30-day and 6-month survival)36
- Significant improvement in NYHA class for 83% of patients36
- Over a 100% improvement in 6-minute walk distance36
- Progressive, sustained improvements in quality of life36
HeartMate 3 also offers:
- A full range of operation, providing flow from 2.5 to 10 L/min to accommodate a broad range of clinical needs
- Artificial pulsatility, designed to promote pump washing and minimize areas of stasis in the pump
- Intrapericardial placement, for surgical ease and minimized complications
- A thin, mechanical apical cuff lock, for quick and easy pump attachment
Important Safety Information
Intended use: HeartMate 3 is intended to provide hemodynamic support in patients with advanced, refractory left ventricular heart failure; either for short-term support, such as a bridge to cardiac transplantation (BTT) or myocardial recovery, or as long-term support, such as destination therapy (DT). HeartMate 3 is intended for use inside or outside the hospital.
Contraindications: HeartMate 3 is contraindicated in patients who cannot tolerate or are allergic to anticoagulation therapy.
Common side effects: Death, bleeding, cardiac arrhythmia, infection, respiratory failure, device malfunction, sepsis, right heart failure, renal failure, stroke, neurologic dysfunction, psychiatric episode, peripheral or device-related thromboembolic event, hemolysis, hepatic dysfunction, myocardial infarction.